Welcome to the Moroni Lab - Biofabrication for Medicine
The Moroni lab develops new biofabrication technologies to generate libraries of 3D scaffolds able to control cell fate. This passes through the design of biomaterials, 3D scaffolds, and surface properties to better understand cell-material interactions.
Current tissue engineering and regenerative medicine products suffer from high costs and laborious techniques that complicate scaling-up production. First generation products consisted of cells in suspension, encapsulated in hydrogels, or seeded into 3D porous matrices. These products demonstrated the potential of regenerative medicine therapies by reducing pain and restoring tissue continuity. Yet, the regenerated tissue is not always as functional as the original one. This leads to degeneration few years after surgery and consequently to the need of another surgery. Causes are different. Cells need to be expanded before achieving a sufficient number for implantation. Cell expansion is typically performed on 2D surfaces, while in the body cell proliferation and homeostasis happens in a 3D environment. This is associated with a loss of the original cell phenotype. Consequently, the expanded cells produce a different extracellular matrix (ECM), ultimately resulting in a tissue formation that is different than the targeted tissue to regenerate. Furthermore, surgical procedures with these products typically consist of two steps, namely isolation and expansion of cells from a tissue biopsy and cell seeding on scaffolds prior to implantation. This is associated with long hospital stay and rehabilitation time, increasing healthcare costs as well.
Our overarching goal is to create new solutions for regenerative medicine and understand the fundamental phenomena at the base of the observed regenerative processes.
Latest News
-
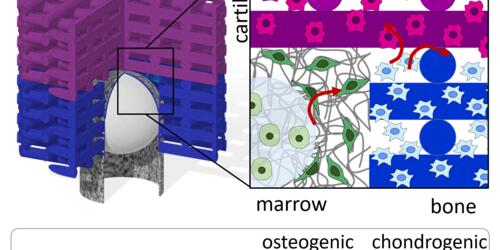
Integrating Biofabrication Technologies
Unlocking the secrets of osteochondral regeneration has long been a daunting challenge, with the intricate interplay between cartilage and bone presenting formidable hurdles. However, a groundbreaking approach is on the horizon, poised to redefine the landscape of tissue regeneration: multiphasic scaffolds – the cornerstone of cutting-edge strategies aimed at mirroring the complexities of the osteochondral unit and nurturing the growth of implanted bone-marrow derived stem cells (BMSCs). Yet, amidst the promise, hurdles remain. Stem cell loss during expansion in vitro and the limited control over their behavior within scaffolds both in vitro and in vivo pose formidable challenges.
-

Bioprinting in Space
In a new Pathfinder Open project, PULSE, we have partnered with different experts in space technology and use thereof to study how the Space environment could be working as an accelerator of ageing…
-
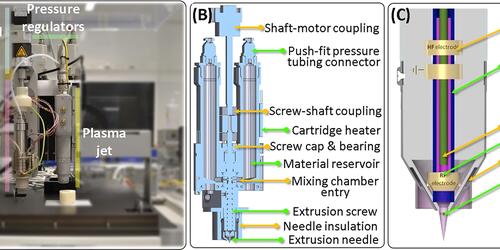
New Hybrid Biofabrication technology
Many tissues in our body display gradients. These are not only biological gradients, but also structural, physical, and chemical ones, resulting in smoother variations of mechanical properties and cell functional activity.
-
Kidney 3D in vitro models through bioprinting
At the Complex Tissue Regeneration department, we work hard to bridge the gap towards the dream of organs bioprinting. Step by step, we are now progressing towards understanding more and more in depth the requirements to bioprint different kidney cells, either derived from pluripotent stem cells or of adult species.
Sponsorship
The major aim of our lab is to develop innovative biofarication approaches for regenerative medicine as well as training next generation's talented students and postdocs.
One of the most direct ways of contributing to these causes is by donating towards a research aim or sponsoring any of our group members directly. Please contact Professor Moroni about donations towards research for fighting diseases such as osteoarthritis, cardiovascular, and neural degeneration.
We are greatful to our generous sponsors!
